Moderate post-therapy efficacy was observed in some of these studies. After infusion, the therapeutic efficacy of the MDDCT is assessed by interrupting ART to check whether viral load suppression can be maintained in the absence of drugs. Moreover, conserved portions of variable antigens such as Env have been considered. HIV-1 peptides typically employed in MDDCT design have been derived from highly conserved proteins of HIV-1, i.e., Gag, Pol, and Nef.
This choice was determined by the presence of a high percentage of defective viruses in cells of patients under long-term ART, rendering difficult the isolation and in-vitro expansion of high amounts of replication-competent HIV-1. While MDDC therapy (MDDCT) has been typically administered to HIV-infected individuals under ART, the virus used for MDDCT design or preparation was obtained from samples of treatment-naive individuals. Peptide-pulsed DCs are then reinfused into the donor to provide an appropriate antigenic stimulus within the body when retroviral replication is still suppressed by ART. ĭC-based therapy relies on in-vitro pulsing or electroporating of autologous monocyte-derived DCs (MDDCs) with whole inactivated HIV-1, with RNA coding for viral antigens, or with specific sets of peptides, to induce recognition of viral epitopes by the individual’s human leukocyte antigens (HLAs) on the DC surface. Due to these characteristics, therapy approaches based on DCs have been proposed and tested to modulate the host’s immune response to HIV-1 infection. DCs can migrate to lymph nodes and other HIV-1 sanctuaries and, once activated, produce interleukins such as IL-12, IL-15, and IL-18, promoting a potent cytotoxic T-cell response necessary for the elimination of infected cells. These cells are anatomically positioned as sentinels for detecting danger signals, thus triggering an innate and adaptive immune response.
Extend flowjo trial professional#
Pivotal for the immune response, dendriticcells (DC) are professional antigen presenters. Of note, the extent of post-therapy viral load rebound is proportional to the size of the HIV-infected cellular reservoir, and the size of this reservoir is in turn proportional to the length of the period during which the infection progresses without ART. However, this response does not typically lead to immunological control of the infection, as shown by the rapid rebound of viremia after interruption of antiretroviral treatment (ART). The onset of HIV-1 infection induces a robust immune response by the host.
Extend flowjo trial registration#
Trial registration NCT02961829: (Multi Interventional Study Exploring HIV-1 Residual Replication: a Step Towards HIV-1 Eradication and Sterilizing Cure,, posted November 11th, 2016) MDDC had a mature profile, and this MDDCT promoted in-vitro T-cell immune responses in HIV-infected patients undergoing long-term suppressive antiretroviral treatment. This was accompanied by an increasing trend in the frequency of polyfunctional T-cells over time, which was visible when considering both cells expressing two and three out of the three cytokines examined. The number of candidates that increased in vitro the cytokine levels in CD4 + and CD8 + T cells upon stimulation with Gag peptides from baseline to day 15 and from baseline to day 30 and day 120 after MDDCT was significant as compared to Gag unstimulated response.
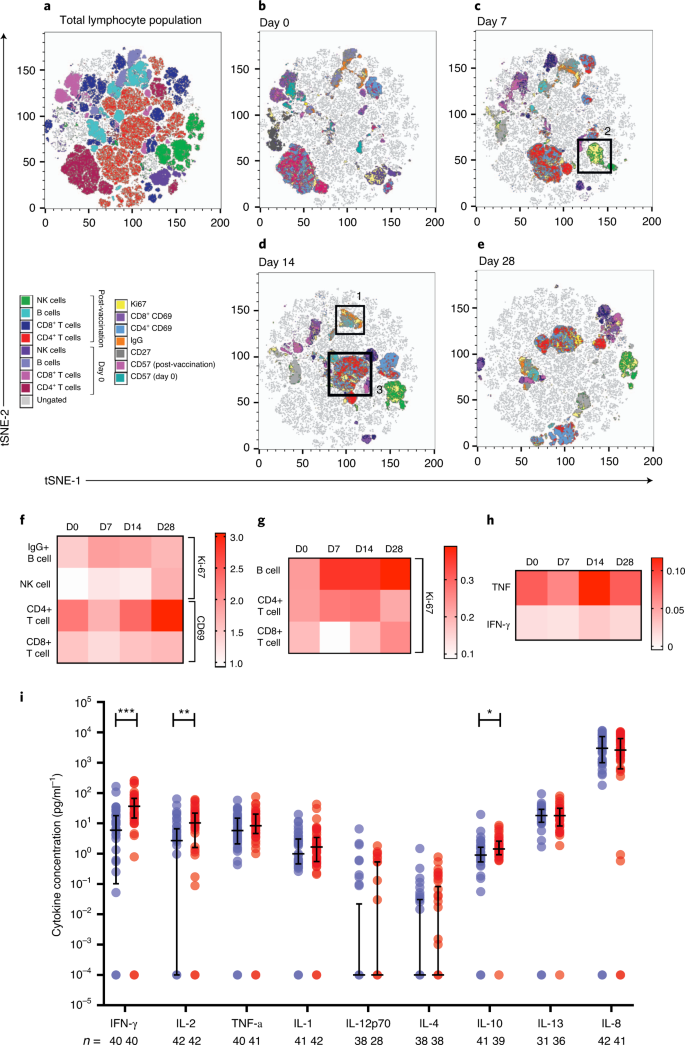
Moreover, intracellular TNF and IFN-γ expression was significantly increased in CD4 + T-cells. MDDCT administration was associated with increased expression of IL-2 in CD4 + and CD8 + T-cells at 15 and/or 30 days after the first MDDCT administration. The protocol of ex-vivo treatment with IFN-α and GM-CSF was able to induce maturation of MDDCs, as well as to preserve their viability for reinfusion. To assess immunogenicity, patients’ cells were stimulated in vitro with autologous peptides, and intracellular IL-2, TNF, and interferon-gamma (IFN-γ) production were measured in CD4 + and CD8 + T-cells. Three doses of the MDDCT were administered every 15 days. After sequencing each patient’s HIV-1 Gag and determining HLA profiles, autologous Gag peptides were selected based on the predicted individual immunogenicity and used to pulse MDDCs. Monocytes were differentiated into DCs using IFN-α and GM-CSF. PBMCs were obtained from 10 HIV + individuals enrolled in trial NCT02961829.

We developed a personalized Monocyte-Derived Dendritic-cell Therapy (MDDCT) for HIV-infected individuals on suppressive antiretroviral treatment and evaluated HIV-specific T-cell responses.
